Ocean Acidification- Laboratory Report
The Role of Carbon Di Oxide On Ocean Acidification
Topic Introduction-
oceans are full of food, transportation, oxygen to provide and regulate climate and also life. When the PH level of these life sources decrease it will higher the rate of acidity in the ocean, which is called ocean acidification. The main factor for ocean acidification is carbon dioxide which can be formed in many ways such as Naturally from us, when we exhale, artificially from gases released from industries and so many other. By different means when it gets in ocean, it raises the level of acidity because when carbon di oxide reacts with water it results carbonic acid CO2+ H20= H2CO3 which is what will happen in the ocean. ocean acidification challenges lives of human, animals and plants in many ways. It results global warming, challenge lives in the ocean, as it becomes acidic, species become extinct and in general messes up the ocean life, which is related to the ecosystem out of water which depends on ocean life and the third party who is dependent on the second party so it affects everything, it also affects shell life, which is also an significant part of life in ocean. Below is a report on the experiment done on the effect of carbon dioxide in ocean acidification and the effect of ocean acidity in shell life.
Experimental Question:
What are we trying to find
out? How?
what effect does carbon di oxide has if it's absorbed in the ocean? We're trying to find out by conducting an experiment.
Pre-Lab Questions: Bubbles
Protocol (answer in complete
sentences)
1. What gas are you blowing into the water?
The gas that was blown in the water was carbon dioxide gas
2. What happens to the gas when you blow it into the water?
when carbon di oxide is blown into the water it is absorbed
3. How are you measuring change in the water during this
lab?
we are measuring the change by timing and recording the change of color
4. What does measuring the pH of the water tell us?
The measure of PH tells us the acidity, neutral or basic characteristics of water
5. After studying the reactions above, how do you think carbonic
acid will affect the pH of salt water?
Carbon dioxide gives carbonic acid when added to water, so carbonic acid will increase the acidity of water and affect it's beings
hypothesis: bubbles
Protocol- when you blow air into sea water, the carbon dioxide you exhale changes the color of the sea water and makes the water acidic.
Hypothesis: Shells
Protocol- when shells are put in vinegar they will get brittle and shrink down while in sea water they stay hard and shiny.
Protocol: Bubble experiment
- The first thing that we did when starting this experiment was assigning role in our group (reader, time keeper and material organizer/bubbler)
- Got all the materials we need including our dependent variable which is the sea water and independent variable which is carbon dioxide (exhaled air)
- Add a 100ML water in the beaker
- using the pipet put 4 drops of universal indicator in the beaker
- wrap the top part with a saran wrap
- poke the saran wrap with your straw and start bubbling for two minutes without stopping
- got the materials we need independent variable being the vinegar and sea water, dependent being the shells
- measure all the shells we have and record the results
- put two of the shells, one in water and one in vinegar
- record the weight afterward
Data Table:
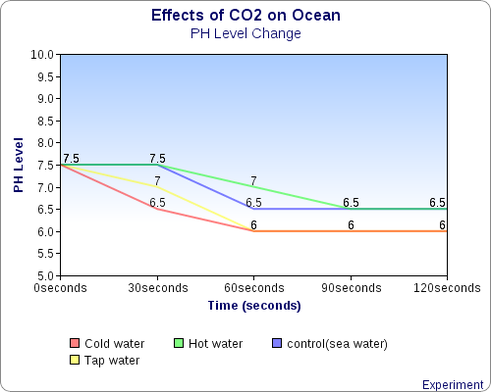
Graph:
Create a graph that includes the data from the bubbles protocol. Use different colors for each of the controls/experimental groups. Make sure to include a key. (see below).
Data Analysis:
(Post-Lab Questions)
1. As you blew through the straw, what were you adding to the
water and how did that change the pH?
As we were blowing in the water, carbon dioxide was being released. And that created carbonic acid when added to water so it will decrease the PH.
2. What did the universal indicator tell us about the water?
The universal indicator shows the color getting lighter/fading, which indicates
decreasing PH level.
3. What does this tell us about the effects of carbonic acid in ocean water?
When carbonic acid is formed in ocean water, it will rise the level of acidity in the
water.
4. Based on the results of your experimental protocol, which factor affects the pH of the water most, temperature or salt?
Temperature
because when the temperature increases the PH level has a little bit more time
to decrease relatively to cold water.
Conclusion: Was your initial hypothesis correct? Why or why not? Explain. What did you learn about
ocean acidification?
The hypothesis "when you blow air into sea water, the carbon dioxide you exhale changes the color of the sea water and makes the water acidic" is proved to be true through this experiment. The experiment we conducted shows that carbon dioxide will create carbonic acid when reacted with water, and will result the decreasing of PH and increases acidity level. From this experiment I noticed that carbon dioxide is the main source of the means of ocean acidification, and will result problem from messing up the ecosystem to extinction of species.
Create a graph that includes the data from the bubbles protocol. Use different colors for each of the controls/experimental groups. Make sure to include a key. (see below).
Data Analysis:
(Post-Lab Questions)
1. As you blew through the straw, what were you adding to the
water and how did that change the pH?
As we were blowing in the water, carbon dioxide was being released. And that created carbonic acid when added to water so it will decrease the PH.
2. What did the universal indicator tell us about the water?
The universal indicator shows the color getting lighter/fading, which indicates
decreasing PH level.
3. What does this tell us about the effects of carbonic acid in ocean water?
When carbonic acid is formed in ocean water, it will rise the level of acidity in the
water.
4. Based on the results of your experimental protocol, which factor affects the pH of the water most, temperature or salt?
Temperature
because when the temperature increases the PH level has a little bit more time
to decrease relatively to cold water.
Conclusion: Was your initial hypothesis correct? Why or why not? Explain. What did you learn about
ocean acidification?
The hypothesis "when you blow air into sea water, the carbon dioxide you exhale changes the color of the sea water and makes the water acidic" is proved to be true through this experiment. The experiment we conducted shows that carbon dioxide will create carbonic acid when reacted with water, and will result the decreasing of PH and increases acidity level. From this experiment I noticed that carbon dioxide is the main source of the means of ocean acidification, and will result problem from messing up the ecosystem to extinction of species.
Central Question:
How does a decrease in the pH of seawater affect the calcium carbonate shells of animals
As the ocean changes it's PH level from an increase in CO2, it is known that the animal life will be impacted by it. As carbon dioxide increases carbonate decreases and carbonate is the main thing in the formation of shell and it dissolve shells which also leads to decreasing of them. Also the animals who make the shells will be affected and cause decrease in their population.
Student Pre- lab
Questions:
1. How do organisms make their shells? What are shells made of?
Organisms make their shells by combining carbonate and calcium molecules.
2. What do you expect to happen to the shell in an acidic solution such as vinegar?
I expect the shell to be brittle and broke down easily related to the normal shells.
3. What are sources of carbon dioxide and which of these sources are most likely to
affect ocean pH?
Sources of carbon dioxide can be humans, industries, burning of coal,through respiration, but the one which affects the ocean are the one's from industries and coal burning because they are more likely to be more artificial and come in a huge that can make a negative impact.
Hypothesis:
If shell is put in a vinegar it would weight less compared to the ones that are in a control environment/place.
Data:
Analysis/Discussion of
Data
1. When you immersed the shells in vinegar how did you know that a reaction was happening?
when we immersed the shell in the vinegar there was small bubbles forming in the tube, which tells us the reaction is happening.
2. How did observing the shells in vinegar relate to how animals are affected by a lower pH of ocean water?
observing shells in a vinegar is related to how animals are affected by ph. level because vinegar has a ph. level of 2 , it's a strong acid, so when the shells dissolve In the vinegar which is acid shows us what happens to the habitats in ocean when ocean is acidic, like a smaller version demonstration of what happens.
3. How would shelled organisms be affected by a lower pH of ocean water?
When the ocean is acidic it slowly leads the shell to dissolve, then shelled organisms wouldn't have a place to stay and be more open up to danger.
4. What are the primary functions of shell for these animals?
They're mainly used for protection as they are made from CaCO3 which is hard material.
5. Does it cost the animal energy to rebuild or repair their shell?
It does cause an amount of energy to rebuild their shell.
Conclusion/summary (revisit hypothesis)
In this lab we placed two shells one in control (sea water) other in vinegar, which is acidic and helps us demonstrate what will happen to the shell when the ocean is acidic. The hypothesis If shell is put in a vinegar it would weight less compared to the ones that are in a control environment/place, is proved to be true due to our data during and after the experiment. It was found that the shells in the vinegar weighed less than what they weighed when we started the experiment. So we conclude that ocean acidification is indeed harmful to dissolve and destroy shells which leads animals who live in shells to danger and decreasing population.
Data
1. When you immersed the shells in vinegar how did you know that a reaction was happening?
when we immersed the shell in the vinegar there was small bubbles forming in the tube, which tells us the reaction is happening.
2. How did observing the shells in vinegar relate to how animals are affected by a lower pH of ocean water?
observing shells in a vinegar is related to how animals are affected by ph. level because vinegar has a ph. level of 2 , it's a strong acid, so when the shells dissolve In the vinegar which is acid shows us what happens to the habitats in ocean when ocean is acidic, like a smaller version demonstration of what happens.
3. How would shelled organisms be affected by a lower pH of ocean water?
When the ocean is acidic it slowly leads the shell to dissolve, then shelled organisms wouldn't have a place to stay and be more open up to danger.
4. What are the primary functions of shell for these animals?
They're mainly used for protection as they are made from CaCO3 which is hard material.
5. Does it cost the animal energy to rebuild or repair their shell?
It does cause an amount of energy to rebuild their shell.
Conclusion/summary (revisit hypothesis)
In this lab we placed two shells one in control (sea water) other in vinegar, which is acidic and helps us demonstrate what will happen to the shell when the ocean is acidic. The hypothesis If shell is put in a vinegar it would weight less compared to the ones that are in a control environment/place, is proved to be true due to our data during and after the experiment. It was found that the shells in the vinegar weighed less than what they weighed when we started the experiment. So we conclude that ocean acidification is indeed harmful to dissolve and destroy shells which leads animals who live in shells to danger and decreasing population.